Merck Animal Health
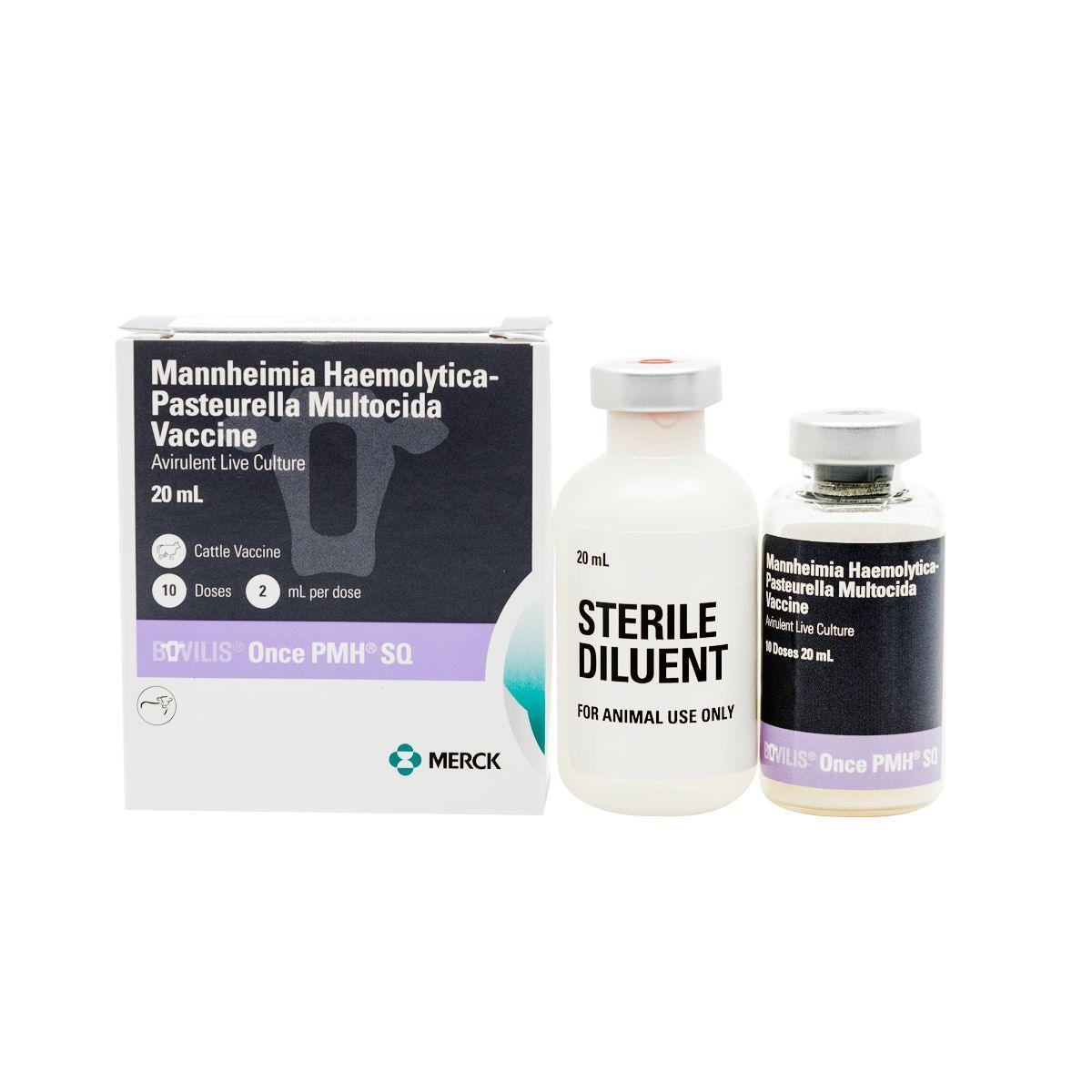
Once PMH SQ
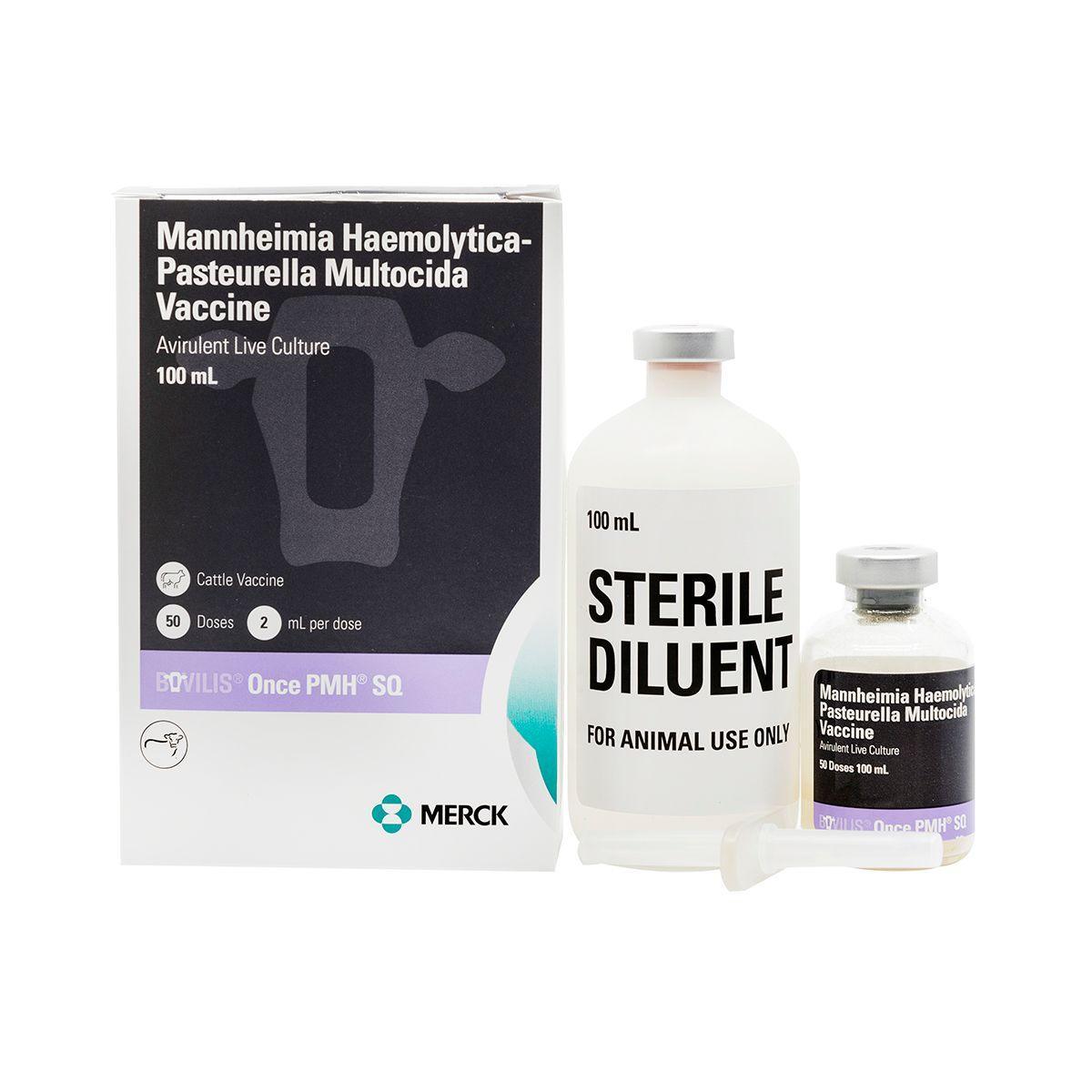
Once PMH SQ
Couldn't load pickup availability
Please shipping information regarding vaccines before placing order.
Once PMH is an avirulent vaccine for healthy cattle to aid in the control of respiratory disease caused by Mannheimia haemolytica and Pastuerella multocida (shipping fever). It’s safe to use on pregnant animals and creates a higher immune response than killed vaccines. A single-shot vaccine for Pasteurella mannheimia and multocida protection of bovine respiratory disease in beef and dairy cattle. For the vaccination of healthy cattle, 3 months of age or older, as an aid in the control of respiratory disease caused by Mannheimia haemolytica and Pasteurella multocida. Calves nursing immune-dams should be vaccinated when maternal antibody levels will allow active immunization
For 10 dose presentation: Rehydrate freeze-dried vial of Once PMH® SQ with accompanying vial of diluent. Mix reconstituted vial well.
For 50 dose presentation: Rehydrate freeze-dried vial of Once PMH SQ® with part of the accompanying vial of diluent using the transfer needle provided (see insert for pictorial directions). Mix reconstituted vial well and transfer rehydrated vaccine into diluent vial using transfer needle. Remove transfer needle from former diluent vial and mix reconstituted vial well. Peel label from bottle of Once PMH SQ® and place on diluent vial containing all vaccine
Administer 2 ml subcutaneously to healthy cattle 3 months of age or older. Calves nursing immune dams should be vaccinated when maternal antibody levels will allow active immunization. Annual revaccination is recommended. A revaccination dose can be administered at more frequent intervals based on farm disease risk assessment or when epidemic conditions exist. Consult your veterinarian
Dosage: 2 mL IM to healthy cattle, revaccinate annually. 21 day slaughter withhold.
Manufacturer and/or Label Information
Manufacturer and/or Label Information
Dimensions
Dimensions
Care information
Care information
Let customers speak for us