Vaxxinova US
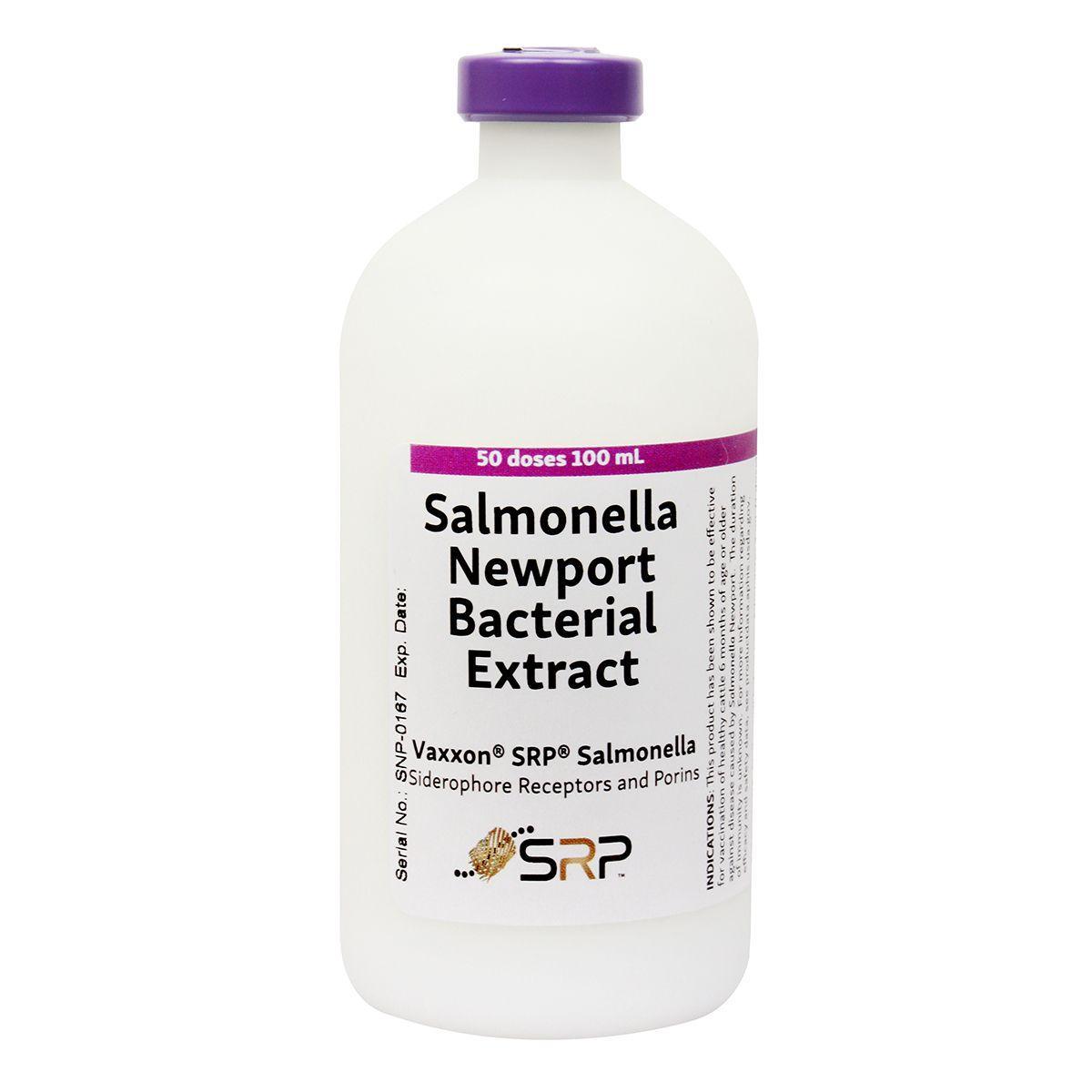
Vaxxon SRP Salmonella-50 Dose
Vaxxon SRP Salmonella-50 Dose
Couldn't load pickup availability
Please see shipping information regarding vaccines before placing order.
For use in healthy cattle 6 months of age or older, as an aid in the control of disease caused by Salmonella Newport. Unique SRP (siderophore receptors and porins) technology creates antibodies that prevent harmful salmonella bacteria from stealing the iron they need to survive from the host animal. Without essential iron, the salmonella die. Helps control new infection, minimizing antibiotic use and discarded milk.
Manufacturer and/or Label Information
Manufacturer and/or Label Information
Dimensions
Dimensions
Care information
Care information
Let customers speak for us